
6
MARKET SPOTLIGHT
FEEDBACK
A D V A N C E D M A T E R I A L S & P R O C E S S E S | M A Y 2 0 1 5
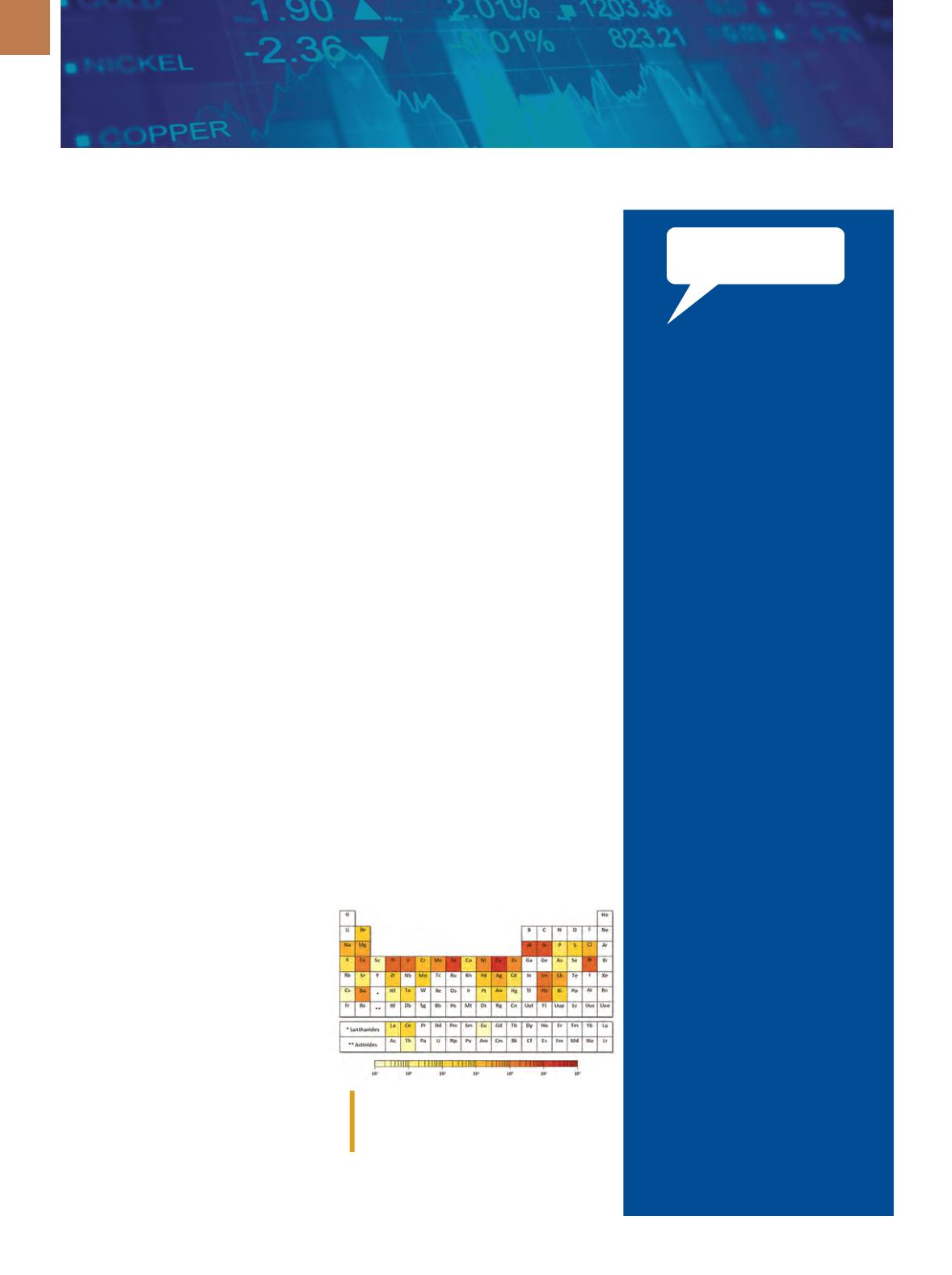
REPORT ASSESSES CRITICALITY OF
ALL METALS ON PERIODIC TABLE
In a new report, researchers from
Yale University, New Haven, Conn., as-
sess the criticality of all 62 metals on the
periodic table, providing key insights
into which materials might become
more difficult to find in the coming de-
cades, which ones will exact the highest
environmental costs, and which ones
cannot be replaced as components of
vital technologies. Many of the metals
traditionally used in manufacturing,
such as zinc, copper, and aluminum,
show no signs of vulnerability. But oth-
er metals critical to the production of
newer technologies like smartphones
may be harder to obtain in coming
decades, says Thomas Graedel, pro-
fessor of industrial ecology at the Yale
School of Forestry & Environmental
Studies. The study represents the first
peer-reviewed assessment of the crit-
icality of all of the planet’s metals and
metalloids.
“The metals we’ve been using for a
long time probably won’t present much
of a challenge,” says Graedel. “But
some metals that have been deployed
for technology only in the last 10 or 20
years are available almost entirely as
byproducts. You can’t mine them spe-
cifically and they don’t have any decent
substitutes.”
According to researchers, critical-
ity depends on more than geological
abundance. Other factors include the
potential for finding effective alter-
natives in production processes, the
degree to which ore deposits are geo-
politically concentrated, the state of
mining technology, regulatory over-
sight, geopolitical initiatives, regional
instabilities, and economic policies.
In order to assess the state of all met-
als, researchers looked at supply risk,
environmental implications, and vul-
nerability to human-imposed supply
restrictions.
Supply limits for many metals crit-
ical to the emerging electronics sector
(including gallium and selenium) are
the result of supply risks. Environmen-
tal implications of mining and process-
ing present the greatest challenges
with platinum-group metals, gold, and
mercury. For steel alloying elements
(including chromium and niobium) and
elements used in high-temperature al-
loys (tungsten and molybdenum), the
greatest vulnerabilities are associated
with supply restrictions.
Among the factors contributing
to criticality challenges are high geo-
political concentration of primary pro-
duction, lack of available substitutes,
and political instability. For example, a
significant portion of tantalum, wide-
ly used in electronics, comes from the
war-ravaged Democratic Republic of
the Congo.
For more information, visit
environment.yale.edu.Yousay ALUMINUM,
I say ALUMINIUM
I recently read the two historical
articles on the Hall-Heroult process
used to make aluminium (“Metallur-
gy Lane,” September and October
2014). It was particularly interesting
to read about Charles Hall’s devel-
opmental work and the evolution of
the appropriately named Pittsburgh
Reduction Co. I did a literature search
several years ago and found that the
word for the element stems from its
presence in the mineral alum. Refin-
ing of the ore produced a derivative
of its name—alumina. The reduction
process resulted in what was initially
called aluminum.
Then, in the late 19th century,
an agreement was reached with the
predecessor of the International
Union of Pure and Applied Chemistry
to add the letter “i” to its name to
bring its spelling into line with the
other elements, e.g., helium, lithium,
and sodium. The icing on the cake to
the “Metallurgy Lane” articles would
have been to answer the question of
why the American Chemical Society,
in 1927, reverted to the 19th century
name for aluminium, i.e., aluminum.
Tony Wells, Australia
[Attention metal history buffs:
If you have a theory or an answer
to this naming puzzle, please drop
us a line.—Eds.]
We welcome all comments and
suggestions. Send letters to
frances. richards@asminternational.org.
Concentrations (in parts per million)
of elements on a printed circuit board.
Courtesy of Thomas Graedel, et al.


















